Study Tracking System
Manage the visits of your study
Join our Partnership Program
With an initial study discount!
Join by clicking on the Apply Now button
Its main characteristics are:
-
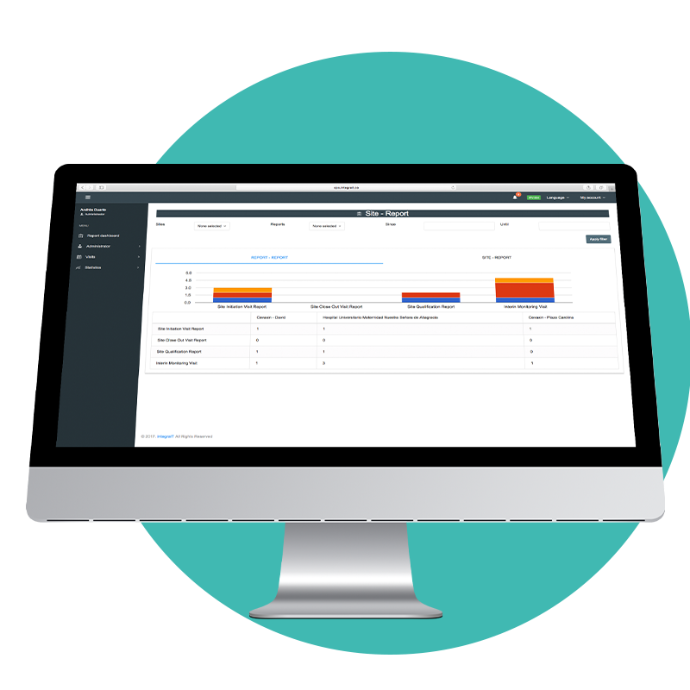
Creation and approval of reports
-
PDF downloads
-
Schedule monitor visits
-
Register and download reports
If you want to know more about how our systems are validated and comply with 21 CFR Part 11
We develop solutions as a service, take a look at how they work.
CONTACT USTypes of Reports
Qualification Visit
Site Initiation Visit
Interim Monitoring Visit
Close-out Visit
Modules
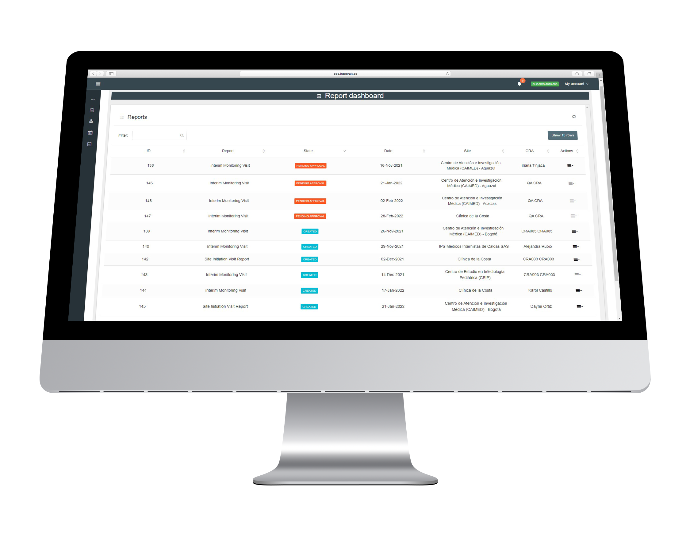
Report Dashboard
This module allow you to see the status of every visit on the project and in what state it is (created, pending approval or approved).
An important view highlights:
-
Protocol Deviations
-
Action Items (AI)
-
SAEs
KPIs with filters per Project, Site, etc. so that the CRO can quickly define next actions.

Visits
This module allows creation and configuration of visits, passing through all the steps needed to collect all the information during all the process, with forms to fill every information needed. Also a calendar is shown with a monthly, weekly or daily view of all the visits in all the studies.
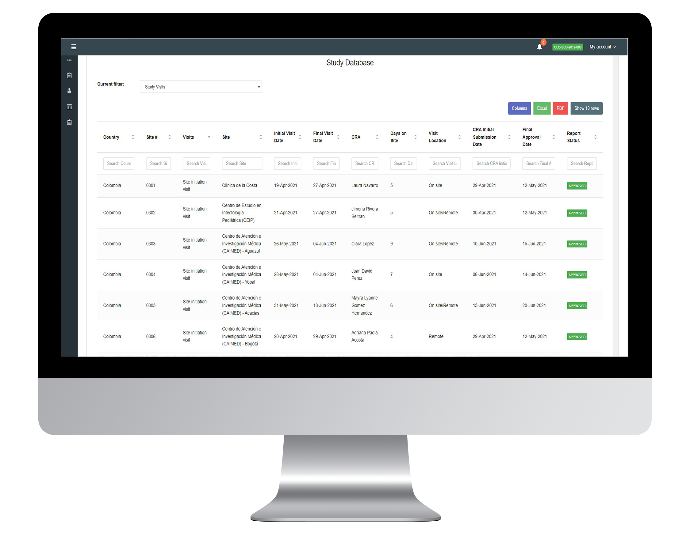
Study Database
The study database module allows the user to see all the information on the database stored with all the visits, including every important information, such as country, site, type of visit, CRA in charge, Status, etc. Letting you download the reports on Excel and PDF.

